Database of luminescent minerals
Ruby (French name: rubis )
Chemical formula: See CORINDON
Family: Oxides and hydroxides
Status: NON APPR
Crystal system : Rhomboedric
Display mineral: OUI
Associated names (luminescent varieties, discredited names, synonyms, etc.): anyolite,
Luminescence:
Longwave UV (365nm) colors: |
Red , | ||
Intensity LW:Strong | |||
Midwave UV (320nm) colors: |
Red , | ||
Intensity MW:Medium | |||
Shortwave UV (254nm) colors: |
Red , | ||
Intensity SW:Medium | |||
Longwave (365nm) picture

Rubis, UV LW,
Luc Yen, Vietnam,
Photo & Col. � G. Barmarin
Pictures Galery:


 ...
...  Go to the galery (10 pictures)
Go to the galery (10 pictures)
Do you have a photo of this mineral you would like to see in the gallery? Contact us!
Phosphorescence (in the common sense of the term) observable with the naked eye:
No phosphorescence visible to the naked eye under any type of UV
Thermoluminescence: OUI
Comments:
Anyolite: corindon in zoïsite and pargasite rock mined in Tanzania. Prilep rubies in the Republic of Macedonia appear to be a mixture of diaspore and corundum. They have the particularity of having an orange fluorescence in some areas and a red (classic) fluorescence in others with rarely also a yellow fluorescence at the top or center of the crystals. Orange fluorescence could be attributed to diaspore (Mn2+ replacing Al3+ with a charge compensation mechanism see diaspore) and ruby red (Cr3+ replacing Al3+). Margarite (green mica) included in corundum emits also a white to slightly greenish fluorescence (see bibliography below) The slight almost linear displacement of the wavelength of the R1 and R2 lines with pressure (several GPa) makes it possible to measure colossal pressures with a certain degree of accuracy.
Activator(s) and spectrum:
Activator(s): Cr3+,
Peaks in the spectrum (nm):
Cr3+ replacing Al3+ : Lines at 692.8 (693), 694.3nm (sharp R1 and R2 lines) Cr3+ : 658.2 , 668.2 , 674.1 , 680 (small lines) N-lines (Cr3+ pairs) : 705.8 , 712nm (small lines)
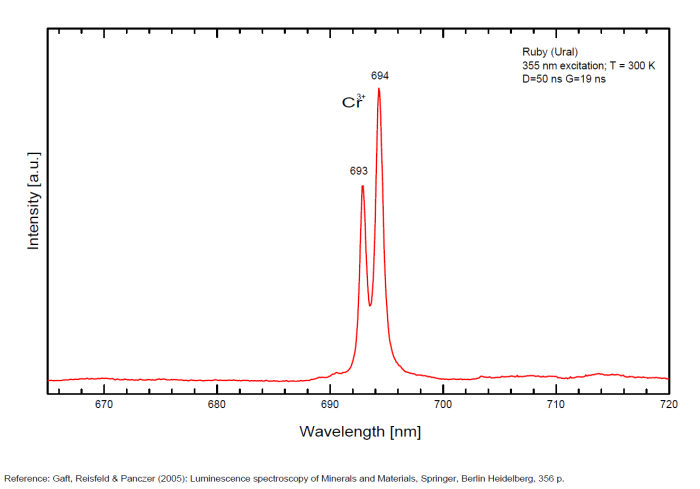
Spectrum: Michael Gaft, Petah Tikva, Israel. Plot: Institute of Mineralogy, University of Vienna, Austria, with permission of the authors.
Spectrum Galery:
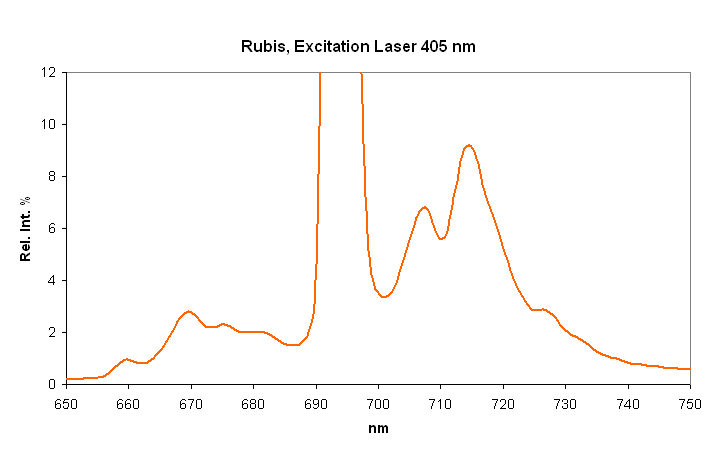
 ...
...Comments on spectrum and activators:
Activator: Cr3+ replacing Al3+ giving strong well known 2E->4A2 lines with long decay time; Besides that, much weaker narrow lines present, which are connected with Cr-pairs and more complicated complexes (so called N-lines) (Tarashchan 1978). Lifetime of the R-line: 3,6ms;
Best localities for fluorescence (*):
- Mogok Township, Pyin-Oo-Lwin District, Mandalay Region, Myanmar;
- Mundarara Mine (Mdarara), Longido, Arusha Region, Tanzania (Anyolite: rubis in zoïsite and pargasite)
- Kleggåsen Ruby Quarry, Froland, Aust-Agder, Norway (strong red LW);
- Hunza Valley, Gilgit, Pakistan;
- Mysore District, Karnataka, India;
- Froland, Norway;
- Fiskenæsset, Nuuk, Sermersooq, Greenland;
- Jegdalek (Jagdalek; Jagdalak; Jagdalik) Ruby Mine, Sorobi District, Kabol (Kabul) Province, Afghanistan;
- Sivec marble quarries, Sivec Mountain, Prilep, Republic of Macedonia (yellow, orange and red fluorescence sometime combined) ;
(*)The data are not exhaustive and are limited to a few remarkable localities for fluorescence
Bibliographic reference for luminescence:
- The Henkel Glossary of Fluorescent Minerals, Dr. Gerhard Henkel, Published by the FMS, 1989 ,
- Fluorescence: Gems and Minerals Under Ultraviolet Light, Manuel Robbins, 1994, Geoscience Press, ISBN 0-945005-13-X ,
- The World of Fluorescent Minerals, Stuart Schneider, Schiffer Publishing, 2006, ISBN 0-7643-2544-2 ,
- Luminescence Spectroscopy of Minerals and Materials, M. Gaft, R. Reisfeld, G. Panczer, Springer Editor, ISBN: 10 3-540-21918-8 ,
- Luminescent Spectra of Minerals, Boris S. Gorobets and Alexandre A. Rogojine, Moscow, 2002 ,
- Ultraviolet Light and Fluorescent Minerals, Th. Warren, S. Gleason, R. Bostwick, et E. Verbeek, 1995, ISBN 0-9635098-0-2 ,
- Luminescenza nel regno minerale, Guido Mazzoleni, fotografia Roberto Appiani, Libri Sandit, 2010, ISBN 978-88-95990-63-7 ,
Reference for luminescence on the Internet:
- Rubis Prilec, Republic of Macedonia: http://fluo.mineralogie.be/SpectraoxidesEng.html#spectrarubyprilebENG
- Diasporescence in Rubies from Prilep Dolomitic Marble, Miha Jeršek, Macedonian Journal of Chemistry and Chemical Engineering, Vol. 34, No. 1, pp. 139–143 (2015)
- Corundum from Prilep Dolomitic Marble (Macedonia), Miha Jeršek1 & Breda MirtiÄ via Palaminerals
Images:
- Froland, Norway: http://www.mindat.org/photo-564716.html
- Republic of Macedonia: https://www.mindat.org/photo-916945.html
- Mogok, Myanmar: https://www.mindat.org/photo-434482.html
- Mysore, India: https://www.mindat.org/photo-1031229.html
Mineralogical reference on the Internet:
 http://www.mindat.org/show.php?name=Ruby
http://www.mindat.org/show.php?name=Ruby
 http://webmineral.com/data/Ruby.shtml
http://webmineral.com/data/Ruby.shtml
Internet Search:
 Image search on 'Google Images'
Image search on 'Google Images'
 Search for documents in all languages on Google
Search for documents in all languages on Google
A request providing no result means only that no such reference exists in the database, but it does not mean that what you are looking for does not exist, just not to our knowledge. If you think you have found an error or omission, please let us know via the contact page being sure to cite the source of information.
