Database of luminescent minerals
CASSITERITE
Chemical formula: SnO2
Family: Oxides and hydroxides
Status: IMA-GP
Crystal system : Tetragonal
Display mineral: NON
Luminescence:
Shortwave UV (254nm) colors: |
Yellow , Yellowish , | ||
Intensity SW:Weak | Frequency SW:Rarely | ||
Do you have a photo of this mineral you would like to see in the gallery? Contact us!
Phosphorescence (in the common sense of the term) observable with the naked eye:
No data
Activator(s) and spectrum:
Activator(s): Sn2+, Sn3+, W,
Peaks in the spectrum (nm):
Sn2+ : very large band at 475nm (200nm half-width) Sn2+ : very large band at 535 - 537nm (140nm half-width) Sn2+ : very large band at 550 nm (140nm half-width)
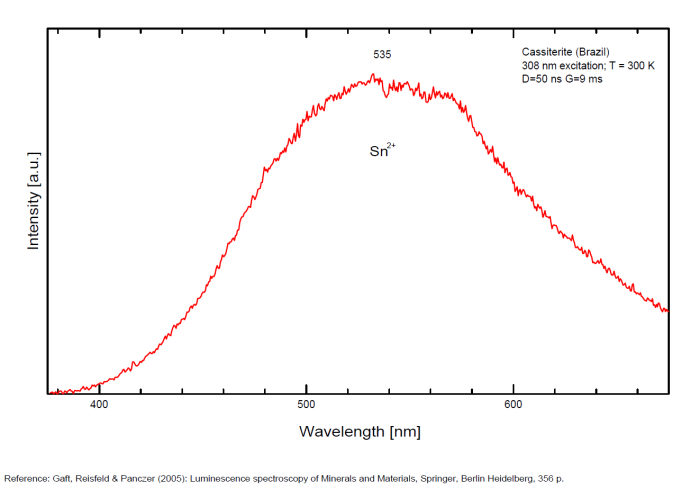
Spectrum: Michael Gaft, Petah Tikva, Israel. Plot: Institute of Mineralogy, University of Vienna, Austria, with permission of the authors.
Spectrum Galery:



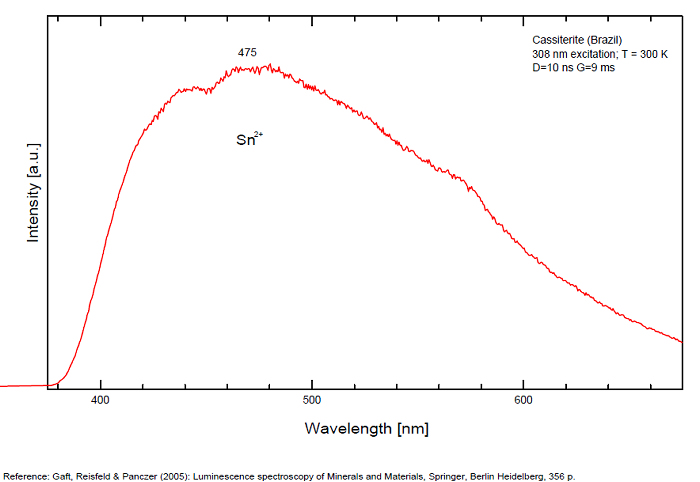 ...
...Comments on spectrum and activators:
Blue luminescence: activator W Yellow luminescence: Ti Under UV lamp excitation cassiterite samples are luminescent only at low temperatures. But with powerful laser excitation (337 nm) even at 300 K or at low temperature (77k) bright intrinsic luminescence (Sn2+ and Sn3+) (Gaft) The spectrum under powerful laser at 300K is similar to those at 77 K with lamp excitation. Decay time of luminescence has two components, namely 100 ns and 280 ns (Gaft and Vorontsova 1982; Gaft et al. 1988).
(Based on Cathodoluminescence, Waychunas)
Best localities for fluorescence (*):
- Aberfoyle Mine (Aberfoyle Tin Mine; Rossarden Tin Mine), Rossarden district, Tasmania, Australia (SW weak yellow);
- Viloco Mine (Araca mine), Loayza Province, La Paz Department, Bolivia (yellow-brown);
(*)The data are not exhaustive and are limited to a few remarkable localities for fluorescence
Bibliographic reference for luminescence:
- The Henkel Glossary of Fluorescent Minerals, Dr. Gerhard Henkel, Published by the FMS, 1989 ,
- Fluorescence: Gems and Minerals Under Ultraviolet Light, Manuel Robbins, 1994, Geoscience Press, ISBN 0-945005-13-X ,
- Luminescence Spectroscopy of Minerals and Materials, M. Gaft, R. Reisfeld, G. Panczer, Springer Editor, ISBN: 10 3-540-21918-8 ,
- Luminescent Spectra of Minerals, Boris S. Gorobets and Alexandre A. Rogojine, Moscow, 2002 ,
Mineralogical reference on the Internet:
 http://www.mindat.org/show.php?name=Cassiterite
http://www.mindat.org/show.php?name=Cassiterite
 http://webmineral.com/data/Cassiterite.shtml
http://webmineral.com/data/Cassiterite.shtml
Internet Search:
 Image search on 'Google Images'
Image search on 'Google Images'
 Search for documents in all languages on Google
Search for documents in all languages on Google
A request providing no result means only that no such reference exists in the database, but it does not mean that what you are looking for does not exist, just not to our knowledge. If you think you have found an error or omission, please let us know via the contact page being sure to cite the source of information.

